Pfizer and the regulators had data by June 2021 showing a serious safety signal for miscarriage. They not only ignored it but suppressed access to the documents.
I have had a number of questions on the subject of miscarriages this week because of a flurry of activity in this area, so I thought it best to put it all in a substack. I hope it becomes a useful reference.
Much of this relates to the pregnancy data from the Pfizer “vaccine” study. You know, the one where they showed how “safe and effective™” the investigational gene therapy vaccine (GTV) products were, particularly in pregnancy.
In fact they are so safe and effective™ in pregnancy that Viki Male – the pharma industry’s go-to for pushing the vaccines on pregnant women – keeps telling us this over and over again. As it happens, Viki is probably not old enough to remember the thalidomide scandal which was one of the many times in history that the pharma lobby has lied to pregnant women about how safe their drug was. The result was a generation of babies that had no limbs, yet this doesn’t stop Viki and the OBGYN establishment (including Kevin Ault, a recent member of ACIP) demonstrating an abject lack of care regarding this completely untested novel therapy.


Adverts for Distaval aka Thalidomide. Women were relentlessly reassured that it was safe in pregnancy, despite grossly inadequate testing. When midwives and doctors tried to raise the alarm they were silenced.
So why am I bringing this up again? Because there are three pieces of information that have come to my attention this week and I need to briefly go over them to show you that the mRNA gene therapy vaccines have had a huge pregnancy safety signal since the beginning of the rollout that should have stopped it in its tracks, but it didn’t.
#1 Pfizer’s PSUR document
This is the Periodic Safety Update Report number 1 (PSUR) which I believe was released under a freedom of information request and was brought to my attention by Sonia Elijah. The safety update report was based on follow up of the Pfizer clinical trial participants as well as separately recorded adverse event reports.

The report itself is 286 pages long but for the purpose of this article we are just going to focus on the pregnancy data pages 234 – 244 (tables 38 – 40). There are a few sources of data quoted but mainly they relate to the C4591001 clinical trial (the “95% effective” study that never was).
This is table 38
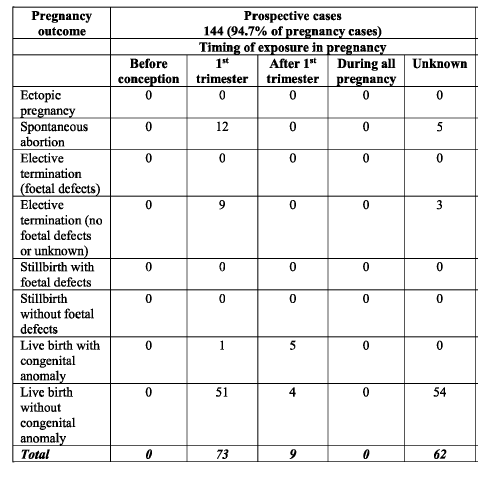
As you can see there were 144 pregnancies recorded prospectively. This is really important because without recording pregnancies prospectively (i.e. from the start) you will miss the pregnancies that miscarry the most – that is those in the first 6 weeks. The V-safe pregnancy registry was meant to have adopted this model but the CDC have chosen not to release that data. I have discussed this previously and for anybody that doesn’t understand how we assess miscarriage rates this article (and the one that it links to) is a must
Igor just blew the lid off the V-safe pregnancy registry …Read more
Now, table 38 and its accompanying data gives us a really useful piece of information that we do not have from any other source – that is the proportion of adverse pregnancy-related events. In this prospective data set 94/145 (65%) of the pregnancies (termed mother cases to avoid the issue of multiple pregnancies) had no adverse event recorded.
In terms of the actual proportion of miscarriages we have the following information
17 of 144 pregnancies (11.8%) miscarried altogether
12 of 73 pregnancies (16.4%) miscarried where the mother received the vaccine in the first trimester
21 of 73 pregnancies (28.7%) miscarried or underwent termination of pregnancy, including those having a termination for fetal defects, where the mother received the vaccine in the first trimester
Now these are shocking figures despite what the pharma companies have told you. What is more they are entirely consistent with what I have written in the previous reports and whichever way you look at it these rates are at least double what you should expect following diagnosis of pregnancy by ultrasound.
Remember as well that it is the first trimester recipients that we are interested in because (a) you can’t miscarry after 20 weeks, a different term is applied and (b) very few drugs have an adverse impact after 20 weeks, even this one.
Just to reiterate also, this data is from the clinical trials and therefore represents a cohort monitored from the time that a pregnancy is declared. Only pregnancies where the outcome was known are included, so the event rates might be higher.
Another shocking figure is this one:
Of the pregnancies recorded, only 76% made it to delivery without a congenital anomaly. 24% ended either in pregnancy loss or congenital anomaly.
It’s also worth noting that in this prospective data set, where “a rigorous assessment was undertaken” as we have been told multiple times, 54 of the 109 live normal births did not have a trimester of exposure recorded. That means that they did not know when the vaccine was given to the pregnant woman.
Now let’s look at table 40.
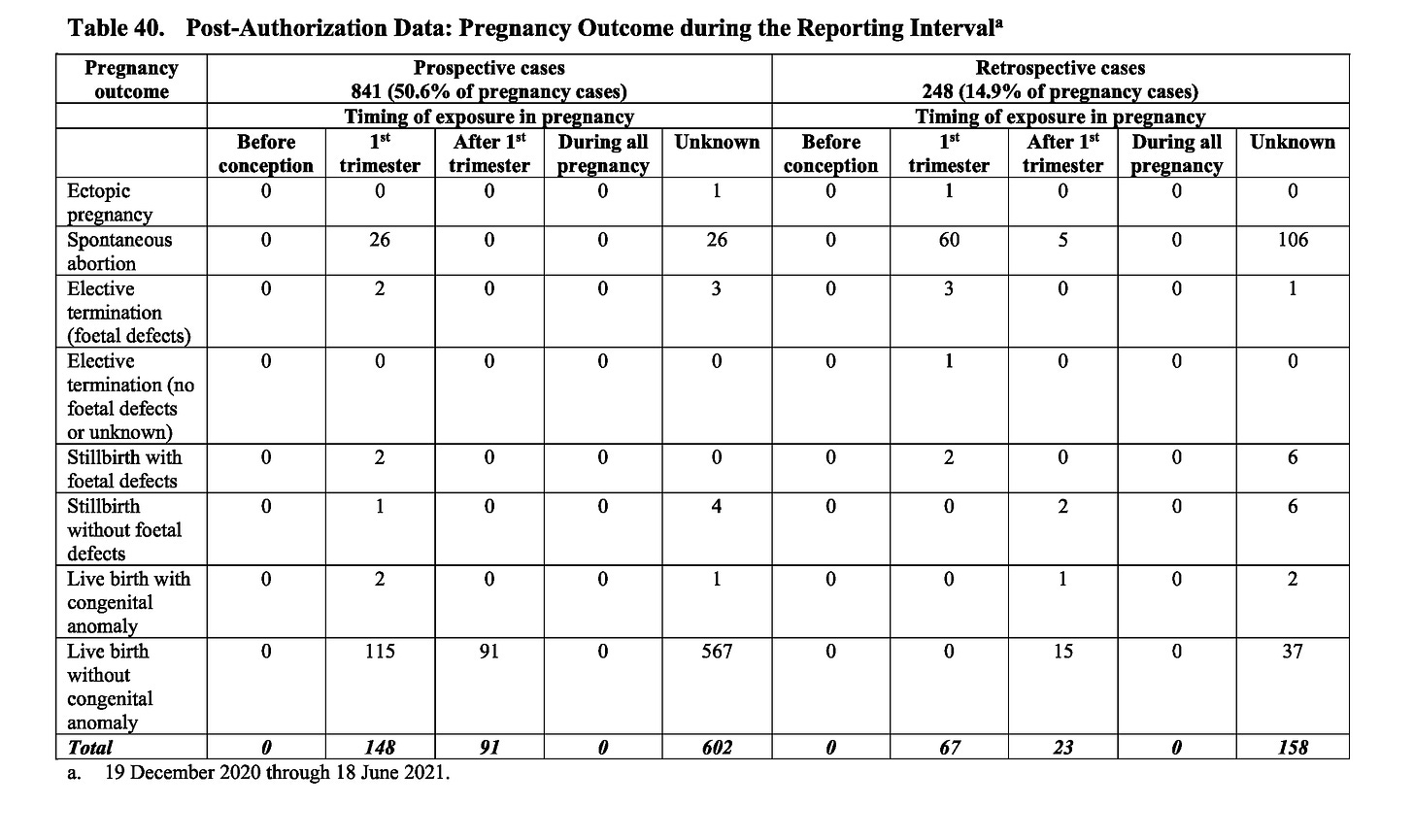
In this table the pertinent figures are these:
Total pregnancies = 1089
Miscarriages* = 232 (21.3%)
First trimester exposure pregnancies = 215
Miscarriages* = 92 (42.7%) [this is an overestimate – see below]*miscarriages and terminations. Note that 90% of the elective terminations in this cohort were for foetal defects – a very high rate. They have therefore been amalgamated with the miscarriages for the purpose of this article.
Now these rates are elevated because the pregnancies recorded in this cohort were from the wider population who reported an “adverse event” as a pregnancy exposure associated with vaccination. So in order to try to ascertain how this population compares to a prospectively evaluated population (where you start off with a known pregnant cohort, give the therapy and then follow them up to completion of pregnancy, just as in V-safe or the table 38 cohort) we need to adjust the denominator upwards.
We can get an estimate of our adjustment factor by using the prospective cohort provided (thanks Pfizer!) in whom the number who reported adverse events (irrespective of the outcome) are recorded. It’s 51/145 (35%). Note that these are the those other than the adverse event of “pregnant on the trial”.

Because this is a prospective cohort we can now use this rate (35%) to reverse engineer the denominator for the larger group in the PSUR, which is in table 40 (the “incremental review” cohort) and is made up of those who reported an adverse event associated with pregnancy relating to receipt of the vaccine – the kind of report you would get in VAERS for instance.

Because this is a selectively reporting cohort, the adverse event rate is higher. This is because some people don’t report (it’s a pain, so why bother?). 659/1604 is only 41% whereas it should be 65%, giving an under-reporting of about 1.5x. So we can predict the denominator such that the reporting rate is 35% to match the clinical trial (“cumulative review”) cohort.
There were 945 reported events (“mother cases”) which should be 35% of the total so the estimated denominator becomes 945/0.35→ 2700
The 1089 total quoted in table 40 was derived from 1607 unique pregnancies (the remainder had not reported) and therefore represents 67.8% of the cohort.
The estimated denominator for table 40 therefore would be 2700*.678→ 1831
On this revised denominator we would then have
Total pregnancies = 1831
Miscarriages* = 232 (12.6%)
First trimester exposure pregnancies (estimated) = 215 *1831/1089 = 361
Miscarriages* = 92 (25.5%)
Note that the calculation is somewhat skewed by the “retrospective cases” in table 40 giving a first trimester miscarriage rate of nearly 100%. This is the same kind of problem that we have seen in previous reports where groups have quoted the adverse event reports with miscarriage rates of nearly 100% – basically because all the reports are of miscarriages after vaccination (this also applies to pretty much every report of a miscarriage rate of over 50%).
Leaving off the “retrospective cases” and only adjusting the “prospective cases” in table 40 (the incremental cohort) gives us
Total pregnancies 841
First trimester exposure miscarriage* = 28/148 = 18.9%
Estimated prospective pregnancies in first trimester = 148*(2700/1609) = 248
Estimated miscarriage rate in first trimester (conservative) = 11.3%
Bear in mind that this is a conservative estimate yet it does come in around the lower limit for the previous estimates of miscarriage rate that we have discussed, and fits in with the Zausche estimate of around 12-14%. The true rate is probably somewhere between the 11% and the 25% calculated. This is up to double that a normal miscarriage rate, whatever Pfizer and the CDC say.
The problem is that, on the basis of the data presented, you would need a much bigger denominator than that seen in the PSUR and this was not provided.
On the basis of the data provided in the PSUR, with miscarriage estimates over 20%, this product should have been halted for pregnant women in the first trimester.
Another way of looking at this is using the text descriptors of the numbers of adverse events

Summarising:
945 pregnancy events. If this is 35% of the population this gives us a revised estimate of 2700 pregnancies
Of the events, 275+21+9+16+7 = 328 are miscarriages (early fetal loss) = 12.1% of the estimated total pregnancies
There is a wide variation in the estimated miscarriage rate, because the data is seemingly deliberately designed to confuse.
There are 9 (not 6) still births reported in the PSUR here1. This gives 0.33%, which is the (lower limit) rate expected for a Western population. This would confirm that the denominator estimate here is probably the upper limit estimate of the correct number.
Assessing the background rate of miscarriage in a normal pregnancy is no mean feat. This is because most early pregnancies and early pregnancy losses are not reported. In order to assess a truly comparative pregnancy cohort you need to recruit participants early and the earliest you can do this is usually around 6-7 weeks of gestation (2-3 weeks after a missed period). “Historical” miscarriage rates always overestimate the miscarriage rate because they include “self reported” pregnancies – which are those not confirmed by ultrasound. Here is an example from a well-touted Lancet review. You can see even from this that the only study that has a miscarriage rate less than 10% is the Andersen study which is a hospital registry. All the other rates are far higher.
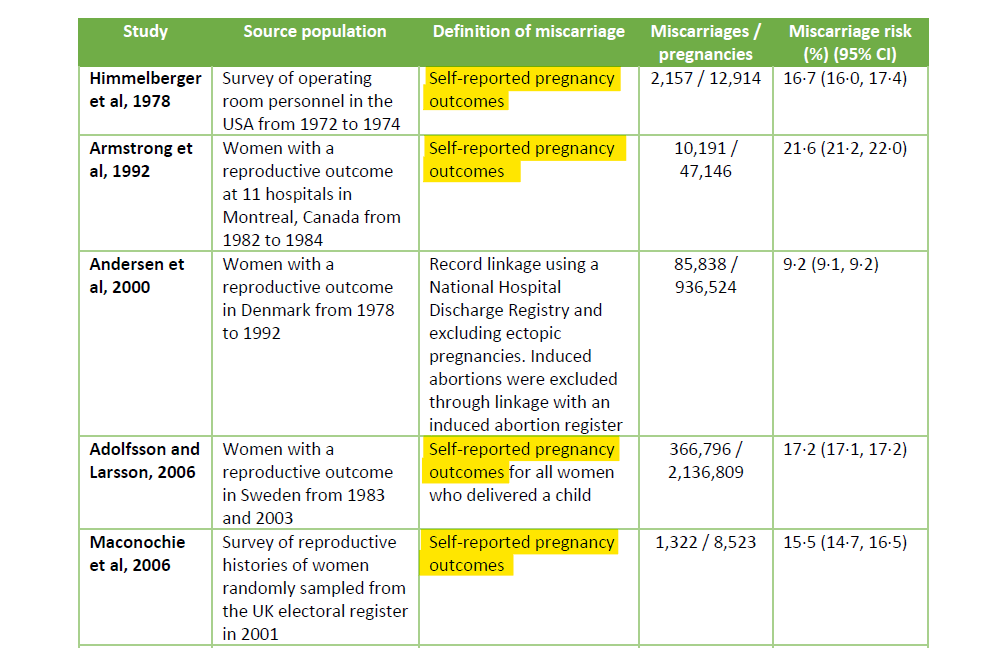
This highlights the difference in comparator when using a prospective cohort. That is, one where you confirm a pregnancy. In a trial situation this is ideally by early ultrasound in order to assess gestational age. Ultrasound is standard practice for dating of pregnancies in most Western countries now, although it wasn’t 20 years ago from which many of the “historical miscarriage rates” are quoted. So the question must be – what is the background rate of confirmed pregnancies now? Papers on this are surprisingly sparse but other than the Naert paper quoted there are two others that show a similar miscarriage rate of 5-6% for ultrasound confirmed pregnancies based on prospective (i.e. women register early in pregnancy, at the latest 6 weeks, and are followed up) – the Wang study here (6/108 miscarriages = 5.6%) and the Bae study here (52/820 miscarriages = 6.3%). If you’re running a trial where you prospectively evaluate patients early on, you need to have a good reason why you aren’t comparing to these rates instead of an inflated rate from 30 years ago.
We now have multiple sources giving us a (conservatively estimated) post-vaccination miscarriage rate of at least 12-16% (prospectively calculated) and multiple sources giving us a pre-vaccination era miscarriage rate of 5-6%. Because a comparative cohort was not provided for V-safe nor for the Pfizer vaccine studies2, it is now impossible to confirm that the product does not increase the risk of miscarriage.
I’ll just repeat that and put it in a box for the TLDR peepsMultiple sources demonstrate that, when prospectively assessed (i.e looking at a cohort of women from the point at which a pregnancy is conclusively demonstrated) the miscarriage rate of a normal cohort is historically 5-6%. Following the mRNA COVID vaccinations multiple sources, presumably using the same criteria, demonstrate miscarriage rates around 12-16%. This represents a potential doubling of the miscarriage rate associated with vaccination in pregnancy.
So now we have the difficult stuff out of the way, we can look at the clues that Pfizer and the regulators (TGA, MHRA, FDA, EMA) knew that there was a problem with fetal loss following the administration of the mRNA products.
#2 The Pfizer-BNT Preclinical studies
Only one animal study was performed in relation to pregnancy by Pfizer-BioNTech before the mRNA vaccine was rolled out, but you knew that didn’t you? In fact the study wasn’t performed by Pfizer itself (as animal studies are a specialised field) but by Charles River, one of the world’s main animal labs used by Pharma.
The document outlining the results of the study was not available to the public until it was eventually forced to be released by two FOI requests to the TGA (2289 and 2389). These are discussed in Maryanne Demasi’s recent expose which I encourage you to read here:
I’m not going to go over the same material as Maryanne but instead I’m going to tell you the story of how we got that data and explain how the regulators (presumably deliberately) missed a massive red flag for pregnancy loss.
Firstly is the story of TGA FOI 2289 which is documented on the Doctors4CovidEthics website.
Essentially the TGA were asked two things:
- Did you see the patient level data?
- Can we see the animal studies?
That was enough to send the TGA into an absolute tailspin of obfuscation and redaction. It turned out that the TGA were hiding the fact that they never saw the patient level data (as demanded also of the FDA in the infamous “you can have it in 55 years” case). In addition, they really did not want to release the animal study data and when it eventually emerged they would only provide a handful of pages of which some looked like this3:

So much for “freedom of information”. What they did allow the release of however was this utopic declaration from the animal studies on fertility – “nothing to see here”. See for yourself.

Just one problem, it was a lie. When the subsequent 2389 document4 was released it was clear that there was a problem. Two problems in fact.
The first problem was that the document was clearly not proofed and edits were left in place. These edits are damning as they indicate that the document was ghost-written (presumably by the sponsor) and simply rubber stamped by the TGA, then rebranded for distribution.

What appears to have happened is that someone told the TGA to downgrade the pregnancy advice category to category B1 (“animal studies showed no problems”) from category B2 (“animal studies not done or inadequate”)
So, were the animal studies adequate and did they show no problems?
Not at all. Firstly, the study was performed on 22 rats in each category (i.e. 22 control rats vs 22 experimental rats). Actually even this wasn’t quite true because the only data available in the main study document shows 21 rats in each arm.
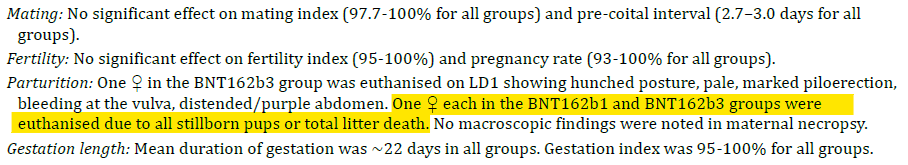
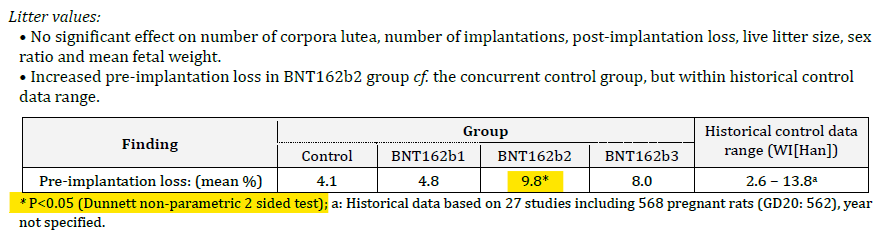
We can reproduce the data from the data from the tables on page 446-457 of the main document. If you look at the “preimplantation loss” column you will see quite a dramatic difference between the groups, with more than a doubling of the losses.
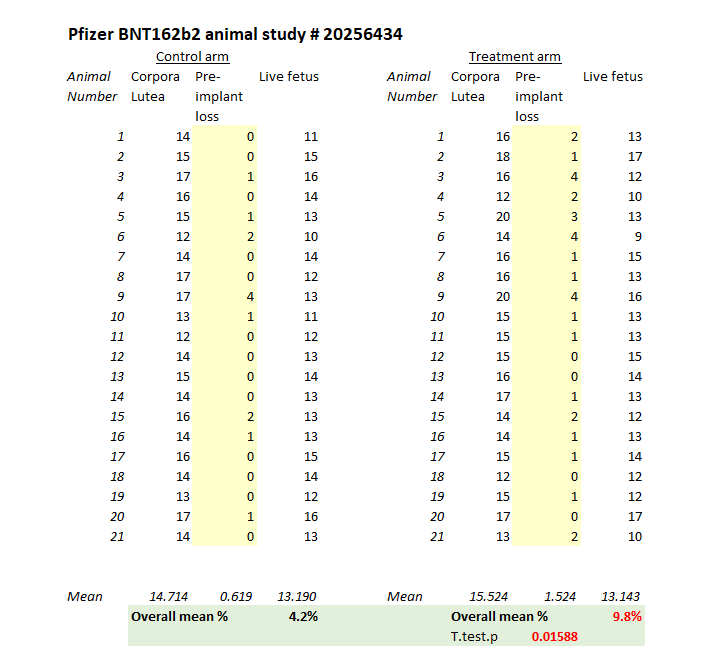
On a straight t-test there is a significant difference between the groups with a p-value of 0.016 (the chance of this result happening randomly is 1.6%) but you wouldn’t do a t-test for this kind of data. You could do a non-parametric test and the usual one that Charles River quoted was the Dunnett test. It’s a glorified non-parametric t-test/ANOVA test for data just like this where you have a control group and then a bunch of other groups to compare against, so it was standard that the study authors used this test.
What did they find? In the BNT162b2 group there was a statistically significant increase in preimplantation losses. This is their data, not mine (although I have replicated it above5). Here is the exact section from document 6 of the TGA FOI 2389:
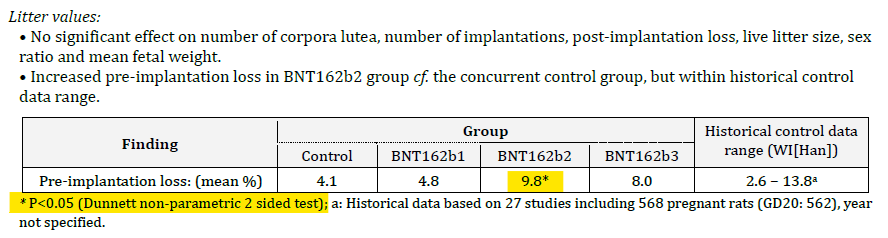
Note that there is another footnote included here with an (a) and directs the reader to look at historical data for comparison. It looks like the Dunnett test was using historical data as a comparator, but this is not the case – the semicolon separates the statements6. It was in fact totally disingenuous to include any reference to historical controls in this table. If you perform a study, your control is your contemporary control (because that controls for the factors that are relevant to the study at the time – each arm being treated equally). The only time you use a historical control is if you are doing a single-arm observational study and don’t have a comparator group.
It would have been OK for the authors to say “we found a higher rate of preimplantation loss in the treatment group, however, this was in the range of historical controls. This would mean that the control group and b1 group had a much lower rate of loss which we can’t explain. We are therefore repeating this experiment”.
Of course they didn’t, because they knew that nobody would care.
The TGA just signed off the document accepting that the study showed a higher rate of preimplantation loss in the animal study and therefore could not be used to justify the “safe” category B1. It didn’t matter because the implication from the document is that they were told to write category B1 and not request any further tests.
Of course the population didn’t care either. Nobody cared that #thalidomideViki was allowed to spout nonsense like this about the very same study which ended up being published after the FOIs meant that it could no longer be kept secret.
Of course the scandal doesn’t stop there. Imagine an “independent research facility” performing “independent animal studies” commissioned by the very company that you (1) need to keep happy to keep their business and (2) are then going to collaborate with to publish your “independent animal study”

What, you thought the animal studies were “independent”? LOL
So, Pfizer-BioNtech not only get to give the TGA and MHRA a soft-soap that the fetal loss rates were double (“yeah but that’s normal”), but they have Viki Male and other stooges running their advertising campaign for them and at the same time they get a free paper out of a pharma-run study, by collaboration with the pharma company. You literally can’t make this shit up.
You’ll be pleased to know that we are at the end of part 2 here and again for the time-poor, here’s a summaryPfizer-BioNtech commissioned Charles River to run animal fertility studies for their product. The TGA tried to suppress the study results. The study showed that the fetal loss rate (a marker for miscarriage/failed pregnancy in humans) was doubled in the treated group and should have prompted further enquiry. The increase was statistically significant on the basis of the test used. Pfizer and Charles River then collaborated to write a paper and claimed that the increased fetal loss rate was normal. The TGA and MHRA rubber stamped the study and nobody asked them to repeat it. This was a major cover-up.
#3 James Thorpe and the VAERS reports
As this has been a long article, I’m very pleased that I don’t have to do much work here because Joel Smalley has done it already. Joel does great work on statistical analysis and if you haven’t subscribed to his substack, you should.
The article is essentially a repost of Dr James Thorpe’s segment on the Tucker Carlson show where he describes his work looking at the VAERS reports of pregnancy-related adverse events compared to other vaccines. VAERS is the FDA’s vaccine adverse event reporting system which is where doctors (mainly) report vaccine adverse events. The Australian equivalent is the DAEN and the UK equivalent is the MHRA yellow card scheme.
As you might expect given this article, there is a massive jump in the number of miscarriages reported to VAERS following COVID “vaccination” when compared to comparison to any other vaccine – specifically the flu vaccine. Most people would consider the flu vaccine a good comparator (it’s not, for reasons I have outlined before, but for the purposes of this comparison it makes sense).
The important segment is shown in Texas Lindsay’s tweet here….
It should be noted that Lindsay means that the 57x increase relates to adverse event reports – not a 57x increased risk of miscarriage (which would be impossible of course). The comparison is to previous VAERS reports and is termed a proportional reporting ratio (PRR). Now, 57 times is a helluva increase and some of it may be due to the under-reporting previously seen with other vaccines (only 1% of adverse events are traditionally reported). The global focus on COVID “vaccines” almost certainly raised the profile of reporting, particularly as the CDC chose to suppress the pregnancy data.
However this increase is not just confined to the US. Another FOI7 to the TGA showed that the TGA were also aware of a large increase in adverse event reporting in relation to miscarriage (aka spontaneous abortion) – of 5.3-fold. Not only was this a dramatic increase in reporting but the TGA’s PRR report also calculates a lower confidence interval, that is the estimate of the lowest possible PRR that the reports could relate to. In other words if the lower limit is more than 1 the increased reporting is likely genuine and needs investigating – particularly if it is more than double. In this case the PRR was more than 5x increased and the TGA ignored it despite meeting its own statistical rules for validity.
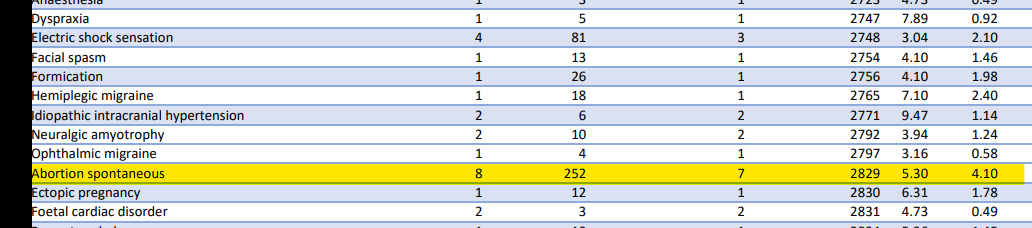
It’s admirable that James Thorpe raises this issue in the public domain, putting his own career at risk. By his own admission it’s statistically a complicated area and he is still finding his feet
In fact, despite being an experienced obstetrician James was one of many to fall into the “50%+ miscarriage” trap laid by the CDC and Pfizer in the way they conflated and confused the public and released dribs and drabs of information on pregnancy. It was a gift for the “fact checkers” to write disparaging articles like this and was the reason I wrote this substack all the way back in September of 2021 – the first year of the rollout:
That article has stood the test of time so far, and was a warning early on that not only was the miscarriage rate likely to be around twice as high following mRNA “vaccination” in early pregnancy8 but that the authorities would do their very best to conflate and cover up.
It’s a travesty that pregnant women – who didn’t benefit from this treatment at all9 – were coerced into taking an investigational product that was known to have a higher rate of fetal loss:
Known before the product came onto the market.
Known at the first “safety” assessment (V-safe)
Known by the FDA and CDC at subsequent safety assessments .
Known by the regulators under proportional reporting ratio warnings.
Known enough that any attempt to get the data and reports was suppressed
Trying to ascertain whether there is a true increase in miscarriage rates is an almost-impossible task because of the wide variation in published rates that absolutely depend on the gestational ages of those assessed. This huge variation produces complete confusion understanding baseline rates and therefore the impact of any intervention. It is the perfect tool with which to hide a medical disaster of the magnitude of the thalidomide scandal, which should have taught us a lesson never ever to forget. The CDC had the chance to recruit an identical control cohort of unvaccinated women in the V-Safe registry but chose not to, as did other groups. The truth may well, in fact, be buried permanently along with the fetal victims of these “studies”. However, on the basis of the available evidence, the COVID “vaccines” should never have been rolled out to pregnant women and should have been paused or halted at the first sign of a safety signal – all the way back in June 2021.
It was, and still is, a miscarriage of medicine.
1 Text from the Pfizer PSUR discussing the stillbirth numbers. The numbers are discordant from those in table 40.

2 Pfizer ran a second trial looking at this question, and then abandoned it.
3 Short and full version of the animal studies as released by the TGA under FOI 2289. The full version was released, a year after the original request, after an official complaint to the information commissioner. Both versions are heavily redacted.
- Foi 2289 Animal Study Short Redacted
- 3.1MB ∙ PDF File – Download
- [d21 3464508] Foi 2289 55g Revised Decision Foi Document
- 19.1MB ∙ PDF File – Download
4 TGA FOI 2389 document 6 (updated version with less redactions)
- Tga Foi 2389 6 Reduced Redations
- 2.52MB ∙ PDF File – Download
5 It was not possible to replicate all four arms of the Dunnett test for this article as the BNT162b1 and BNT163b3 data was redacted. Hence a t-test p-value was provided as an approximation.
6 You wouldn’t use a Dunnett test for a comparison against historical controls. Dunnett is used for a multi-arm contemporaneous study. If a Dunnett test had been used in this experiment the authors could not have found there to be a significant different at p<0.05 and therefore the first part of the statement (“p<0.05 (Dunnett…”) would be nonsensical. I suspect it was intentional that the statement was written this way, to sow confusion amongst those who don’t pay attention (like the TGA and MHRA).
7 Freedom of information request
8 In case you have missed it, miscarriages can only happen in early pregnancy. After 20 weeks any fetal loss is not usually termed a miscarriage at all. After 14 weeks the miscarriage rate – even in the presence of insults such as novel drug therapies – is very low. The fetus is surprisingly robust after 14 weeks. It is therefore disingenuous to use pregnancies after 14 weeks as a denominator when calculating miscarriage rates.
9 An extensive debunking of the “no increased risk of miscarriage” fallacy promoted relentlessly by Viki Male, Kevin Ault and others can be found in the tweet thread referenced earlier in the article (unroll on threadreader here)
Source – https://arkmedic.substack.com/p/the-miscarriage-of-medicine
